A) heat is not a form of energy.
B) cells do not have much heat; they are relatively cool.
C) temperature is usually uniform throughout a cell.
D) heat can never be used to do work.
E) heat must remain constant during work.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How might an amino acid change at a site distant from the active site of the enzyme alter the enzyme's substrate specificity?
A) by changing the enzyme's stability
B) by changing the enzyme's location in the cell
C) by changing the shape of the protein
D) by changing the enzyme's pH optimum
E) an amino acid change away from the active site cannot alter the enzyme's substrate specificity
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
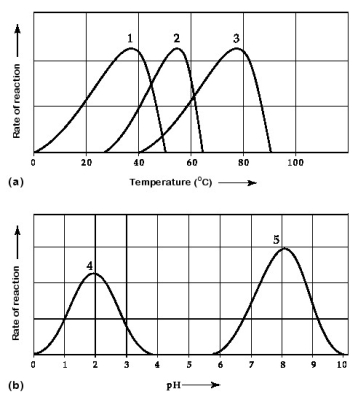 Activity of various enzymes at various temperatures (a) and at various pH (b) .
-Which curve(s) on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
Activity of various enzymes at various temperatures (a) and at various pH (b) .
-Which curve(s) on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
A) curves 1 and 5
B) curves 2 and 4
C) curves 2 and 5
D) curves 3 and 4
E) curves 3 and 5
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following questions are based on the reaction A + B ↔ C + D shown in Figure 8.1.
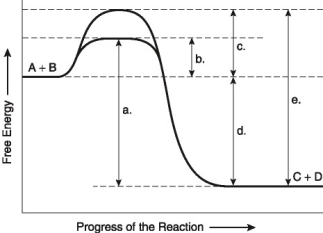 Figure 8.1
-Which of the following represents the activation energy needed for the enzyme-catalyzed reverse reaction, C + D → A + B, in Figure 8.1?
Figure 8.1
-Which of the following represents the activation energy needed for the enzyme-catalyzed reverse reaction, C + D → A + B, in Figure 8.1?
A) a
B) b
C) c
D) d
E) e
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Succinate dehydrogenase catalyzes the conversion of succinate to fumarate. The reaction is inhibited by malonic acid, which resembles succinate but cannot be acted upon by succinate dehydrogenase. Increasing the ratio of succinate to malonic acid reduces the inhibitory effect of malonic acid. -Based on this information, which of the following is correct?
A) Succinate dehydrogenase is the enzyme, and fumarate is the substrate.
B) Succinate dehydrogenase is the enzyme, and malonic acid is the substrate.
C) Succinate is the substrate, and fumarate is the product.
D) Fumarate is the product, and malonic acid is a noncompetitive inhibitor.
E) Malonic acid is the product, and fumarate is a competitive inhibitor.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When 10,000 molecules of ATP are hydrolyzed to ADP and 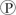 ᵢin a test tube, about twice as much heat is liberated as when a cell hydrolyzes the same amount of ATP. Which of the following is the best explanation for this observation?
ᵢin a test tube, about twice as much heat is liberated as when a cell hydrolyzes the same amount of ATP. Which of the following is the best explanation for this observation?
A) Cells are open systems, but a test tube is a closed system.
B) Cells are less efficient at heat production than nonliving systems.
C) The hydrolysis of ATP in a cell produces different chemical products than does the reaction in a test tube.
D) The reaction in cells must be catalyzed by enzymes, but the reaction in a test tube does not need enzymes.
E) Reactant and product concentrations in the test tube are different from those in the cell.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following questions are based on the reaction A + B ↔ C + D shown in Figure 8.1.
 Figure 8.1
-Which of the following represents the activation energy required for a noncatalyzed reaction in Figure 8.1?
Figure 8.1
-Which of the following represents the activation energy required for a noncatalyzed reaction in Figure 8.1?
A) a
B) b
C) c
D) d
E) e
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An important group of peripheral membrane proteins are enzymes such as the phospholipases that cleave the head groups of phospholipids. What properties must these enzymes exhibit?
A) resistance to degradation
B) independence from cofactor interaction
C) water solubility
D) lipid solubility
E) membrane-spanning domains
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In experimental tests of enzyme evolution, where a gene encoding an enzyme is subjected to multiple cycles of random mutagenesis and selection for altered substrate specificity, the resulting enzyme had multiple amino acid changes associated with altered substrate specificity. Where in the enzyme were these amino acid changes located?
A) only in the active site
B) only in the active site or near the active site
C) in or near the active site and at surface sites away from the active site
D) only at surface sites away from the active site
E) only in the hydrophobic interior of the folded protein
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chemical equilibrium is relatively rare in living cells. Which of the following could be an example of a reaction at chemical equilibrium in a cell?
A) a reaction in which the free energy at equilibrium is higher than the energy content at any point away from equilibrium
B) a chemical reaction in which the entropy change in the reaction is just balanced by an opposite entropy change in the cell's surroundings
C) an endergonic reaction in an active metabolic pathway where the energy for that reaction is supplied only by heat from the environment
D) a chemical reaction in which both the reactants and products are not being produced or used in any active metabolic pathway
E) no possibility of having chemical equilibrium in any living cell
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an example of potential rather than kinetic energy?
A) the muscle contractions of a person mowing grass
B) water rushing over Niagara Falls
C) light flashes emitted by a firefly
D) a molecule of glucose
E) the flight of an insect foraging for food
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mathematical expression for the change in free energy of a system is ΔG =ΔH - TΔS. Which of the following is (are) correct?
A) ΔS is the change in enthalpy, a measure of randomness.
B) ΔH is the change in entropy, the energy available to do work.
C) ΔG is the change in free energy.
D) T is the temperature in degrees Celsius.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A series of enzymes catalyze the reaction X → Y → Z → A. Product A binds to the enzyme that converts X to Y at a position remote from its active site. This binding decreases the activity of the enzyme. -With respect to the enzyme that converts X to Y, substance A functions as
A) a coenzyme.
B) an allosteric inhibitor.
C) the substrate.
D) an intermediate.
E) a competitive inhibitor.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following questions are from the end-of-chapter "Test Your Understanding" section in Chapter 8 of the textbook. -Which of the following metabolic processes can occur without a net influx of energy from some other process?
A) ADP + ![]() ᵢ → ATP + H₂O
ᵢ → ATP + H₂O
B) C₆H₁₂O₆ + 6 O₂ → 6 CO₂ + 6 H₂O
C) 6 CO₂ + 6 H₂O → C₆H₁₂O₆ + 6 O₂
D) amino acids → protein
E) glucose + fructose → sucrose
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true concerning catabolic pathways?
A) They combine molecules into more energy-rich molecules.
B) They supply energy, primarily in the form of ATP, for the cell's work.
C) They are endergonic.
D) They are spontaneous and do not need enzyme catalysis.
E) They build up complex molecules such as protein from simpler compounds.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following questions are based on the reaction A + B ↔ C + D shown in Figure 8.1.
 Figure 8.1
-Which of the following represents the difference between the free-energy content of the reaction and the free-energy content of the products in Figure 8.1?
Figure 8.1
-Which of the following represents the difference between the free-energy content of the reaction and the free-energy content of the products in Figure 8.1?
A) a
B) b
C) c
D) d
E) e
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an example of cooperativity?
A) the binding of an end product of a metabolic pathway to the first enzyme that acts in the pathway
B) one enzyme in a metabolic pathway passing its product to act as a substrate for the next enzyme in the pathway
C) a molecule binding at one unit of a tetramer, allowing faster binding at each of the other three
D) the effect of increasing temperature on the rate of an enzymatic reaction
E) binding of an ATP molecule along with one of the substrate molecules in an active site
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Some of the drugs used to treat HIV patients are competitive inhibitors of the HIV reverse transcriptase enzyme. Unfortunately, the high mutation rate of HIV means that the virus rapidly acquires mutations with amino acid changes that make them resistant to these competitive inhibitors. Where in the reverse transcriptase enzyme would such amino acid changes most likely occur in drug-resistant viruses?
A) in or near the active site
B) at an allosteric site
C) at a cofactor binding site
D) in regions of the protein that determine packaging into the virus capsid
E) such mutations could occur anywhere with equal probability
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order to attach a particular amino acid to the tRNA molecule that will transport it, an enzyme, an aminoacyl-tRNA synthetase, is required, along with ATP. Initially, the enzyme has an active site for ATP and another for the amino acid, but it is not able to attach the tRNA. What must occur in order for the final attachment to occur?
A) The ATP must first have to attach to the tRNA.
B) The binding of the first two molecules must cause a 3-D change that opens another active site on the enzyme.
C) The ATP must be hydrolyzed to allow the amino acid to bind to the synthetase.
D) The tRNA molecule must have to alter its shape in order to be able to fit into the active site with the other two molecules.
E) The 3' end of the tRNA must have to be cleaved before it can have an attached amino acid.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During a laboratory experiment, you discover that an enzyme-catalyzed reaction has a ∆G of -20 kcal/mol. If you double the amount of enzyme in the reaction, what will be the ∆G for the new reaction?
A) -40 kcal/mol
B) -20 kcal/mol
C) 0 kcal/mol
D) +20 kcal/mol
E) +40 kcal/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 80
Related Exams