Correct Answer

verified
Steno demonstrated that the angles betwe...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What charge does the silicate ion contribute to form the silicate tetrahedron?
A) negative
B) no charge
C) positive
D) negative and positive
E) all of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Igneous rocks form from the cooling and solidification of magma.Sample (A,B,or C) _____ has formed by this processes.
A) ![]()
B) ![]()
C) ![]()
E) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Silicon is the most abundant element in Earth's crust.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of weak bond results from asymmetry in charge distribution?
A) covalent bond
B) metallic bond
C) ionic bond
D) Van der Waals bond
F) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
In ______,two elements are so similar in size and bonding properties that one can exchange for the other during crystallization.
Correct Answer

verified
Correct Answer
verified
Short Answer
The _____ of a mineral is the color of the mineral when powdered,which is usually accomplished in soft minerals by rubbing the sample against an unglazed porcelain plate.
Correct Answer

verified
streak
Correct Answer
verified
Multiple Choice
What type of bond forms from the sharing of electrons between atoms?
A) covalent bond
B) metallic bond
C) ionic bond
D) Van der Waals bond
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
An atom that has an excess positive or negative electrical charge caused by the loss or addition of one or more electron is called a(n)______.
Correct Answer

verified
ion
Correct Answer
verified
Essay
What is the difference between ionic and covalent bonding? Which type of bonding creates compounds that tend to be relatively strong and hard?
Correct Answer

verified
Ionic bonding involves electron transfer...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
The illustration below shows five different silicate structures.Of these,the structure labeled _____ is the most abundant in silicate minerals of Earth's crust.
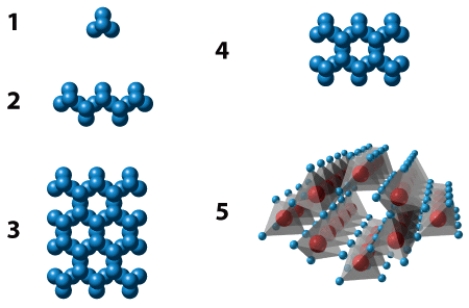
Correct Answer

verified
Correct Answer
verified
Essay
Two chemical elements make up 70 percent of Earth's crust by weight.What are the two elements,and what family of minerals do they form?
Correct Answer

verified
The two elements are...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Based on their origins,rocks can be divided into three distinct families: _______,sedimentary,and metamorphic.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is steel not considered a mineral?
A) It is organic.
B) It is man-made.
C) It doesn't have a crystalline structure.
D) All of the above
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
With approximately 3,500 known minerals,why are there only about thirty common rock-forming minerals?
Correct Answer

verified
The number of rock-forming minerals is l...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
The property of ______ is a mineral's resistance to scratching.
Correct Answer

verified
hardness
Correct Answer
verified
Multiple Choice
Which of the following is the hardest silicate structure?
A) single tetrahedron
B) hexagonal ring
C) sheet
D) framework
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the illustration below,the part of the atom that has a positive charge is referred to as the: 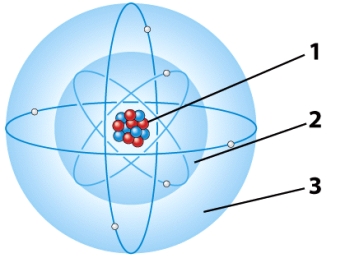
A) first electron energy level.
B) second electron energy level.
C) Both A and B are correct.
D) nucleus.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In the photograph of the quartz crystal below,the angles between similar faces are constant because of the chemical composition of the mineral.

B) False
Correct Answer

verified
Correct Answer
verified
Essay
What is the relationship between cleavage,crystal structure,and crystal faces? Speculate about how different types of bonding might influence each of these.
Correct Answer

verified
Cleavage in some minerals and crystal fo...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 1 - 20 of 75
Related Exams