A) Processes that are spontaneous in one direction are spontaneous in the opposite direction.
B) Processes are spontaneous because they occur at an observable rate.
C) Spontaneity can depend on the temperature.
D) All of the statements are true.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔH° for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen, SO2 (g) → S (s,rhombic) + O2 (g) Is ________ kJ/mol.
A) 0.0
B) +135.0
C) -135.90
D) -269.9
E) +269.9
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
With thermodynamics,one cannot determine ________.
A) the speed of a reaction
B) the direction of a spontaneous reaction
C) the extent of a reaction
D) the value of the equilibrium constant
E) the temperature at which a reaction will be spontaneous
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔH° for the decomposition of POCl3 into its constituent elements, 2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g) Is ________ kJ/mol.
A) -1228.7
B) +1228.7
C) -940.1
D) +940.1
E) 0.00
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
ΔS is positive for the reaction ________.
A) Pb(NO3) 2 (aq) + 2KI(aq) → PbI2 (s) + 2KNO3 (aq)
B) 2H2O (g) → 2H2 (g) + O2 (g)
C) H2O (g) → H2O (s)
D) NO (g) + O2 (g) → NO2 (g)
E) Ag+ (aq) + Cl- (aq) → AgCl (s)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the largest entropy?
A) B2F4 (g)
B) BF3 (g)
C) B2H6 (g)
D) BCl3 (l)
E) B2O3 (s)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The normal boiling point of water is 100.0 °C and its molar enthalpy of vaporization is 40.67 kJ/mol.What is the change in entropy in the system in J/K when 24.7 grams of steam at 1 atm condenses to a liquid at the normal boiling point?
A) 88.8
B) -88.8
C) -150
D) 373
E) -40.7
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: Ag+ (aq) + Cl- (aq) → AgCl (s)
Given the following table of thermodynamic data,  determine the temperature (in °C) above which the reaction is nonspontaneous under standard conditions.
determine the temperature (in °C) above which the reaction is nonspontaneous under standard conditions.
A) 1230
B) 150
C) 432
D) 133
E) 1640
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔG° for a reaction conducted at 25 °C is 3.05 kJ/mol.The equilibrium constant for a reaction is ________ at this temperature.
A) 0.292
B) -4.20
C) 0.320
D) -1.13
E) More information is needed.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔG° at 261.0 °C for the formation of phosphorous trichloride from its constituent elements, 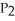 (g) +
(g) + 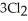 (g) →
(g) → 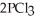 (g) is ________ kJ/mol.At 25.0 °C for this reaction,ΔH° is -720.5 kJ/mol,ΔG° is
(g) is ________ kJ/mol.At 25.0 °C for this reaction,ΔH° is -720.5 kJ/mol,ΔG° is 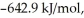 and
and 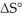 is
is 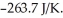
A) -579.6
B) 6.81 × ![]()
C) 1.40 × 105
D) -651.7
E) -861.4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: FeO (s) + Fe (s) + O2 (g) → Fe2O3 (s)
Given the following table of thermodynamic data, 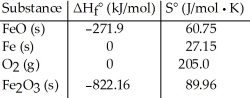 determine the temperature (in °C) at which the reaction is nonspontaneous.
determine the temperature (in °C) at which the reaction is nonspontaneous.
A) below 618.1
B) above 2438
C) above 756.3
D) below 2438
E) This reaction is spontaneous at all temperatures.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant for the following reaction is 3.0 × 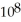 at 25 °C.
at 25 °C. 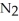 (g) + 3
(g) + 3 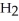 (g)
(g)  2N
2N 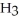 (g) The value of ΔG° for this reaction is ________ kJ/mol.
(g) The value of ΔG° for this reaction is ________ kJ/mol.
A) 22
B) -4.1
C) 4.1
D) -48
E) -22
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
At what temperature (in K)will a reaction become spontaneous? ΔH is 115.0 kJ/mol and ΔS is 75.00 J/K ∙ mol.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔS° for the decomposition of calcium chloride into its constituent elements, CaCl2 (s) → Ca (s) + Cl2 (g) Is ________ J/K ∙ mol.
A) -104.6
B) +104.6
C) +369.0
D) -159.8
E) +159.8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔG° at 25 °C for the decomposition of POCl3 into its constituent elements, 2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g) Is ________ kJ/mol.
A) -1108.7
B) +1108.7
C) -606.2
D) +606.2
E) -1,005
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following processes produces a decrease in the entropy of the system?
A) freezing of Fe(l) into Fe(s)
B) evaporation of liquid ethanol into gaseous ethanol
C) dissolution of LiOH(s) in water
D) melting ice to form water
E) mixing of two gases into one container
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the largest entropy?
A) HI (g)
B) HBr (g)
C) HCl (g)
D) HCl (s)
E) HCl (l)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔH° for the formation of POCl3 from its constituent elements, P2 (g) + O2 (g) + 3Cl2 (g) → 2POCl3 (g) Is ________ kJ/mol.
A) -1228.7
B) -397.7
C) -686.5
D) +1228.7
E) +686.5
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the equilibrium constant for a reaction at 25 °C.The value of ΔG° is -57.5 kJ/mol.
A) 8.4 × 10101
B) 10
C) 1.0
D) 1.2 × 1010
E) more information is needed
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reversible process is one that ________.
A) can be reversed with no net change in either system or surroundings
B) happens spontaneously
C) is spontaneous in both directions
D) must be carried out at low temperature
E) must be carried out at high temperature
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 125
Related Exams