A) I
B) II
C) III
D) IV
E) Both I and II
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which alkene would you expect to have the smallest heat of hydrogenation?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The LUMO of 1,3-pentadiene has how many electrons in its ground state?
A) 1
B) 2
C) 3
D) 4
E) 0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these dienes is the most reactive in the Diels-Alder reaction?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the structure of the conjugated diene. 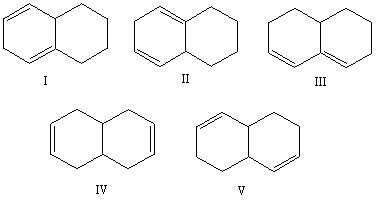
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
What combination of diene and dieneophile can be used to prepare the following compound through a Diels-Alder cycloaddition reaction: 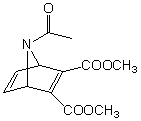
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would afford a synthesis of the following compound? 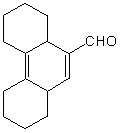
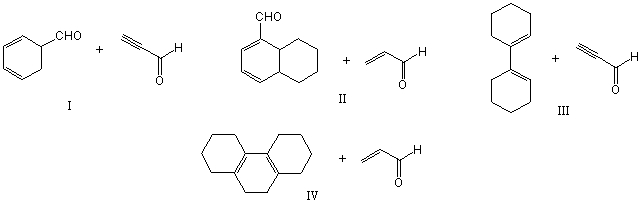
A) I
B) II
C) III
D) IV
E) None of these choices.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the most energetically favorable UV transition for 1,3-butadiene.
A) "n *"
B) "n *"
C) " 2 3*"
D) " *"
E) " 1 4*"
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these dienes can undergo the Diels-Alder reaction?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the structure of the isolated diene. 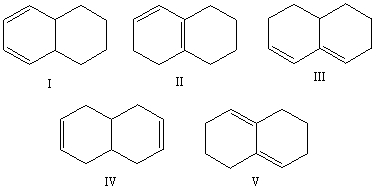
A) I
B) II
C) III
D) IV
E) V
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would afford a synthesis of the following compound? 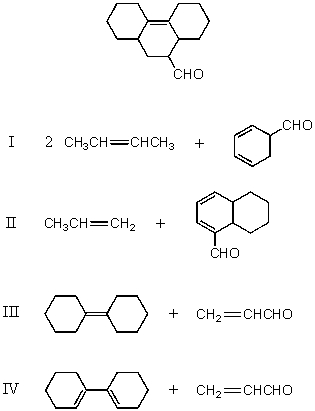
A) I
B) II
C) III
D) IV
E) None of these choices.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Which diene and dienophile would you use to prepare the following molecule using a Diels-Alder cycloaddition reaction: 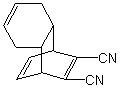
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these dienes can undergo a Diels-Alder reaction? 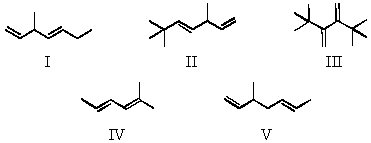
A) I and V
B) I,II and V
C) III
D) IV
E) III and IV
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these dienes can undergo the Diels-Alder reaction?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) All of these choices can undergo the Diels-Alder reaction.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Polyenes in which single and double bonds alternate along the carbon chain are called ___.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The allyl radical has how many electrons in bonding molecular orbitals?
A) 1
B) 2
C) 3
D) 4
E) 5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of compounds could be used as the basis for a Diels-Alder synthesis of the compound shown below? 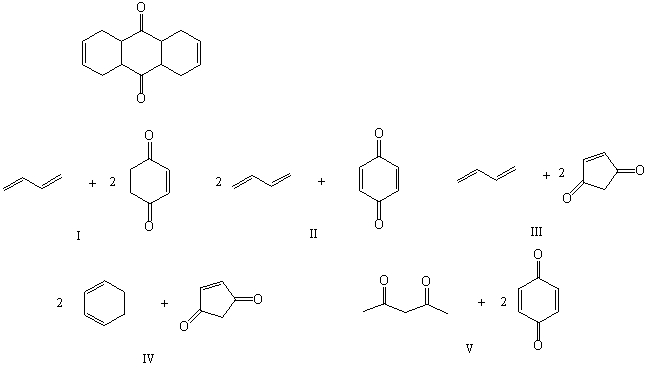
A) I
B) II
C) III
D) IV
E) V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Complete the following sequence of reactions,giving structural details of all key intermediates. 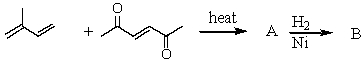
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which diene would be least stable? 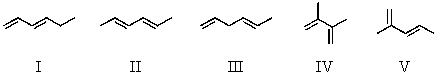
A) I
B) II
C) III
D) IV
E) V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Show the steps and reagents necessary to prepare the following compound from organic compounds having six carbons or less.Use retrosynthetic analysis as a tool to guide your synthesis. 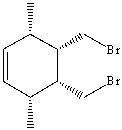
Correct Answer

verified
Correct Answer
verified
Showing 121 - 140 of 201
Related Exams